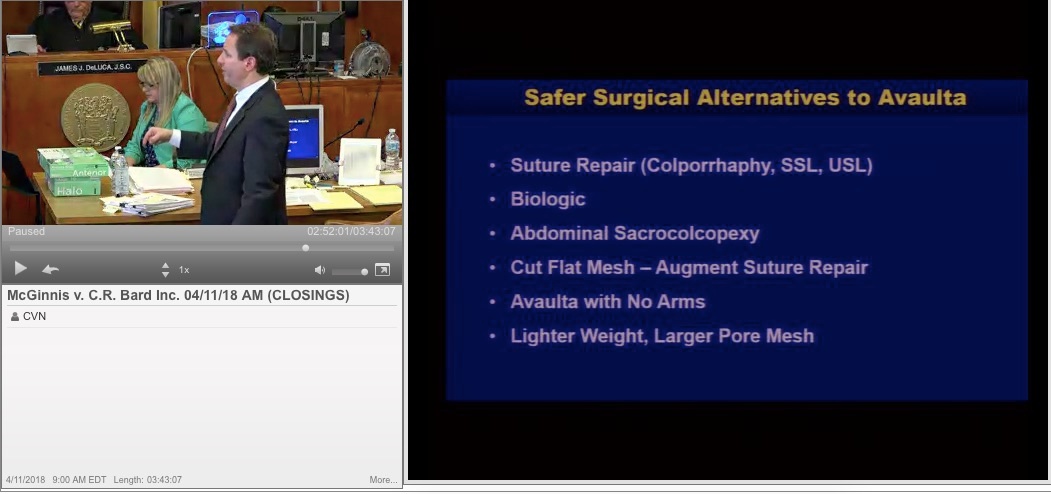
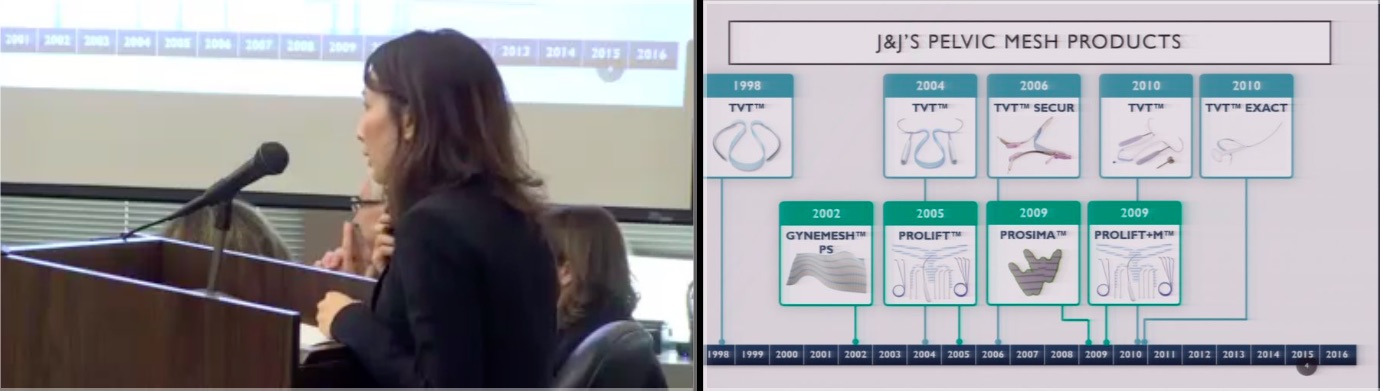
A Summary of Pelvic Mesh Trials in CVN’s Online Video Library
Posted by David Siegel on Apr 16, 2020 2:20:07 PM
Topics: Products Liability, Pelvic Mesh Litigation, California, new jersey, Missouri, Practice Area Overview
Judge Orders J&J To Pay $344M For Deceptive Marketing Of Pelvic Mesh Implants
Posted by David Siegel on Jan 31, 2020 1:57:11 PM
Topics: Pelvic Mesh Litigation, California
California AG’s Office Asks Judge To Award $800M As Nation’s 1st Pelvic Mesh False Marketing Trial Concludes
Posted by David Siegel on Sep 27, 2019 12:57:31 PM
Topics: Products Liability, Pelvic Mesh Litigation, California
Trial In California’s $1B Pelvic Mesh Lawsuit Against Johnson & Johnson Gets Underway
Posted by David Siegel on Jul 19, 2019 3:06:50 PM
Topics: Products Liability, Pelvic Mesh Litigation, California
CVN To Webcast $6M+ Boston Scientific Pelvic Mesh Trial In Massachusetts
Posted by David Siegel on May 6, 2016 8:38:10 PM
Update 5/9/2016: Parties settled the case prior to Monday's opening statements. Terms of the settlement were not disclosed. Neither the parties' attorneys nor Boston Scientific representatives could immediately be reached for comment.
Topics: Products Liability, Pelvic Mesh Litigation