CVN screenshot of California Deputy Attorney General Jinsook Ohta delivering her opening statement
San Diego, CA - Lawyers from the office of the California Attorney General concluded their presentation of evidence on Monday in a long-running trial over the alleged false marketing of pelvic mesh devices manufactured by Johnson & Johnson subsidiary Ethicon.
For the last five weeks lawyers from the AG’s office presented testimony to San Diego County Superior Court Judge Eddie Sturgeon, who is hearing the case without a jury. It is the first case in the country to go to trial involving pelvic mesh-related claims filed by a state as opposed to an actual injured mesh patient.
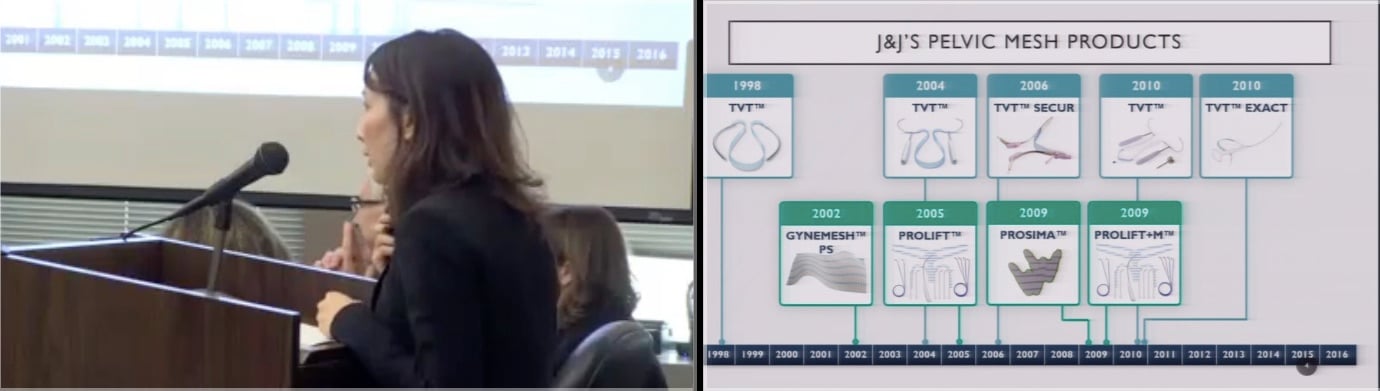
The case alleges Ethicon deceived up to 50,000 California women and doctors into using mesh devices to treat incontinence and organ prolapse without adequately warning them about risks supposedly associated the implants. Depending on how Judge Sturgeon decides the case, Ethicon, which denies the allegations, could be potentially liable for nearly $1 billion in damages.
The full trial, expected to conclude in late September following the presentation of Ethicon’s case is being webcast and recorded gavel-to-gavel by Courtroom View Network.
Thousands of women who received mesh implants manufactured by Ethicon and other companies not named in the current case allege the devices left them suffering from chronic pain, persistent infections and sexual dysfunction. Their claims against Ethicon argue the company knew the chemicals in the mesh could cause an adverse reaction when exposed to human tissue.
CVN recorded a number of those trials in states throughout the country, and gavel-to-gavel video of those proceedings can be accessed by CVN subscribers.
California’s lawsuit accuses the company of failing to adequately inform the public of those supposed risks.
“J&J deceptively marketed its surgical mesh devices as safe with minimal risk when in fact these devices exposed women to a host of dangerous complications,” the AG’s office writes in their complaint. “J&J did this despite being urged by its own medical advisers and employees to warn doctors and patients of pain with intercourse, sexual dysfunction, and impact on quality of life.”
Ethicon and other mesh manufacturers have argued their devices helped thousands of women and offered a safer alternative to invasive surgeries with large incisions that could themselves present a host of life-threatening side effects. They frequently argue the side effects women attribute to mesh implants are actually the result of other pre-existing conditions.
While continuing to defend the safety of their mesh devices in court, Ethicon and other mesh manufactures have voluntarily withdrawn their pelvic mesh products from US markets in recent years.
In April the U.S. Food & Drug Administration ordered the only two companies still selling mesh products for transvaginal use, Boston Scientific and Coloplast, to halt their distribution citing safety concerns.
Two weeks prior to that decision, a lawsuit brought by Washington State similar to the pending California action settled on the eve of trial, with Ethicon agreeing to pay $9.9 million while not having to admit any wrongdoing.
The California AG’s office is relying on a team of in-house attorneys.
Johnson & Johnson and Ethicon is represented by O’Melveny & Myers LLP and Covington & Burling LLP.
The case is captioned The People of the State of California v. Johnson & Johnson, case number 37-2016-00017229-CU-MC-CTL in the Superior Court of the State of California, County of San Diego.





